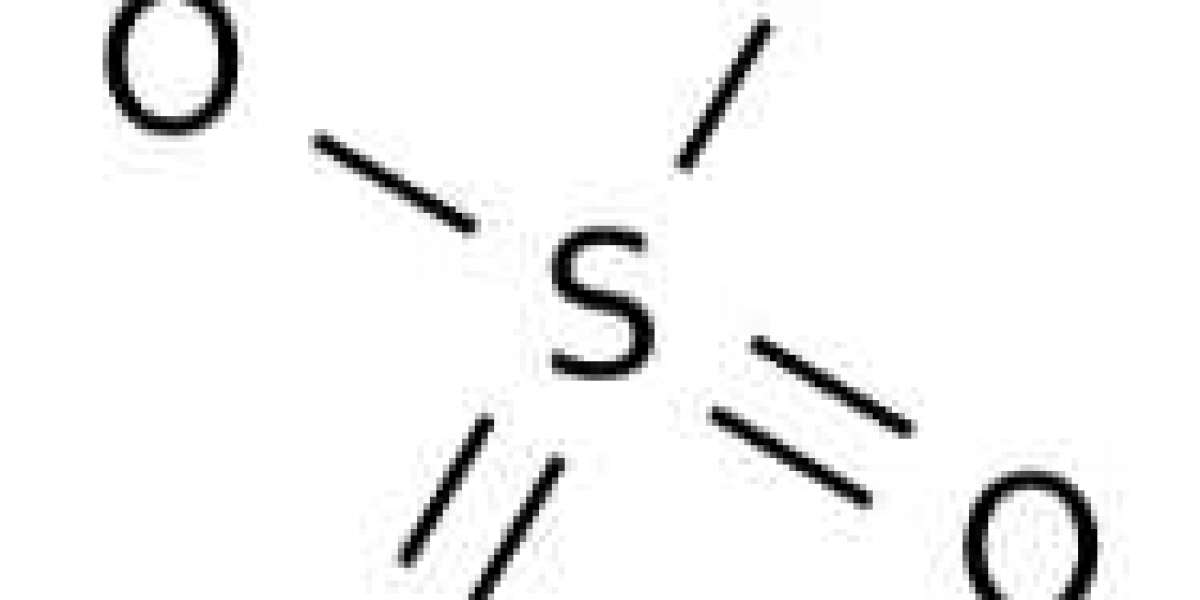
Sulfuric acid is mainly used in fertilizer processing, such as calcium superphosphate and ammonium sulfate. It is commonly used in chemical processing, such as in the manufacture of hydrochloric acid, nitric acid, sulfates, synthetic detergents, dyes and pigments, explosives and pharmaceuticals.
What is the strongest acid in the world?
Carborane superacid can be considered the strongest solar acid in the world because fluoroantimonic acid is actually a mixture of hydrofluoric acid and antimony pentafluorofluorocarbons.
How do you neutralize sulfuric acid?
Pour the baking soda directly into the spilled acid. This will neutralize light acids, such as vinegar, and even toxic strong acids, such as hydrochloric and sulfuric acid. Spray the entire contaminated area with baking soda (sodium bicarbonate, NaHCO3) to neutralize the acid.
Can you use water and sulfuric acid?
Sulfuric acid cannot be mixed with water, but it can be diluted with water. Carefully add a few drops of acid to the water and stir constantly, as the reaction is exothermic. To neutralize sulfuric acid, a strong base like sodium hydroxide can be used.
Is sulfuric acid ionic or molecular?
Water forms a compromise between H2O and H3O+ and -OH. It's not going to produce any ions. Sulfuric acid is a covalent compound because all the bonds are covalent. But it ionizes in solution to form hydronium ions and sulfate ions.
Sulfuric acid is a strong mineral acid.