3-Pentanone is a natural product that occurs in Cichorium endivia, Zingiber mioga, and other organisms for which data are available.
Diethyl ketone is a clear, colorless liquid with an acetone-like odor. Flash point 55°F. Density is less than water. Vapor is heavier than air.
Pentan-3-one is a pentanone, which is pentane with an oxo group in the 3-position. It has been isolated from Triatoma brasiliensis and Triatoma infestans. It has an effect as an animal metabolite.
3-Pentanone (also known as diethylketone) is a simple, symmetrical dialkylketone. It is a colorless liquid ketone with an acetone-like odor. Soluble in about 25 parts of water, but miscible with organic solvents.
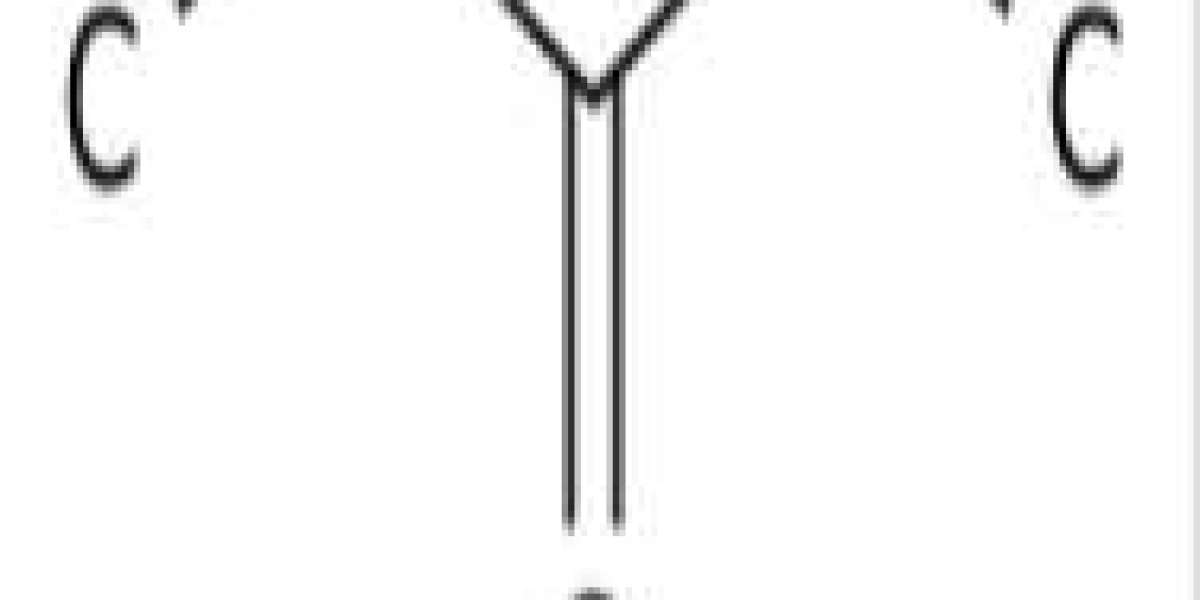
3-Pentanone is produced by the ketodecarboxylation of propionic acid using a metal oxide catalyst:
2 CH3CH2CO2H → (CH3CH2)2CO + CO2 + H2O
In the laboratory, the reaction can be performed in a tube furnace. [4]
Carbonylation route
It can also be prepared by combining ethylene, CO, and H2. [3] Water can be used as a source of hydrogen when the reaction is catalyzed by dicobalt octacarbonyl. The proposed intermediate is the ethylene-propionyl species [CH3C(O)Co(CO)3(ethylene)], which undergoes migratory insertion to form [CH3COCH2CH2Co(CO)3]. The required hydrogen comes from the water shift reaction. For details, see [5] If the water shift reaction does not work, this reaction provides polymers containing alternating carbon monoxide and ethylene units. Such aliphatic polyketones are usually prepared using palladium catalysts. [6]